Current strategies for delivery of macromolecules, such as nanoparticles, liposomes, viral-based vectors, microinjection, and electroporation (Kang et al., 2016; Shi et al., 2018; Hao et al., 2019; Yang et al., 2019b; Yang et al., 2019c), may result in high toxicity, poor specificity, immunogenicity, as well as low delivery efficiency and efficacy (Swain et al., 2016). Different from other delivery strategies mentioned above, CPPs can enter the cells in a noninvasive way, as they usually do not disturb the structure of the plasma membranes and are considered safe and highly efficient. CPPs was first introduced by two research groups in 1988 (Frankel and Pabo, 1988; Green and Loewenstein, 1988). Both Frankel et al. and Green et al. observed that the HIV-trans-activator of transcription (TAT) protein could enter tissue-cultured cells, target into cell nucleus, and finally result in target gene expression. In 1991, Joliot et al. revealed that the homeodomain of Antennapedia (pAntp), a synthetic peptide with 60 amino acids long, was internalized by nerve cells (Joliot et al., 1991). Subsequently, Derossi et al. found a short peptide with 16 amino acids from the third helix of the antennapedia homeodomain (RQIKIYFQNRRMKWKK), named penetratin, was able to penetrate the plasma membrane (Derossi et al., 1994).
Currently, CPPsite 2.0 (http://crdd.osdd.net/raghava/cppsite/) database contains about 1850 kinds of CPP sequences, and the number is expected to continue to increase.
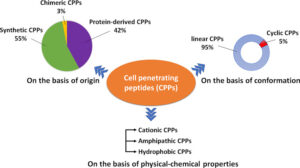
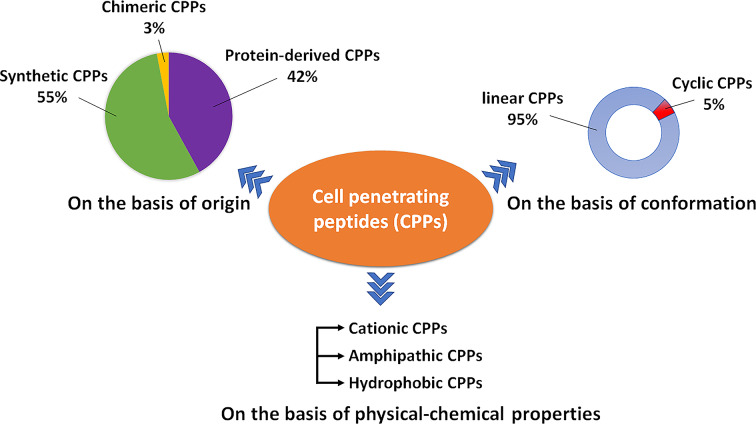
Classification of the types of CPPs.
| Peptide | Sequence | Length | Origin | References |
|---|---|---|---|---|
| Cationic CPPs | ||||
| TAT | RKKRRQRRR | 9 | Protein derived | (Baoum et al., 2012) |
| R8 | RRRRRRRR | 8 | Synthetic | (Chu et al., 2015) |
| DPV3 | RKKRRRESRKKRRRES | 16 | Protein derived | (De coupade et al., 2005) |
| DPV6 | GRPRESGKKRKRKRLKP | 17 | Protein derived | (De coupade et al., 2005) |
| Penetratin | RQIKIWFQNRRMKWKK | 16 | Protein derived | (Nielsen et al., 2014) |
| R9-TAT | GRRRRRRRRRPPQ | 13 | Protein derived | (Futaki et al., 2001) |
| Amphipathic CPPs | ||||
| pVEC | LLIILRRRIRKQAHAHSK | 18 | Protein derived | (Eggimann et al., 2014) |
| ARF (19-31) | RVRVFVVHIPRLT | 13 | Protein derived | (Johansson et al., 2008) |
| MPG | GALFLGFLGAAGSTMGAWSQPKKKRKV | 27 | Chimeric | (Simeoni, 2003) |
| MAP | KLALKLALKALKAALKLA | 18 | Synthetic | (Wada et al., 2013) |
| Transportan | GWTLNSAGYLLGKINLKALAALAKKIL | 27 | Protein derived | (Pae et al., 2014) |
| Hydrophobic CPPs | ||||
| Bip4 | VSALK | 5 | Protein derived | (Gomez et al., 2010) |
| C105Y | CSIPPEVKFNPFVYLI | 16 | Protein derived | (Rhee and Davis, 2006) |
| Melittin | GIGAVLKVLTTGLPALISWIKRKRQQ | 26 | Protein derived | (Hou et al., 2013) |
| gH625 | HGLASTLTRWAHYNALIRAF | 20 | Protein derived | (Galdiero et al., 2015) |
The poly-arginine stretches have the highest cell uptake capacity and have therapeutic potential. The results of the study show that the internalization capacity of oligoarginine increases with its length (Chu et al., 2015), but for delivery purposes, the optimal length is R8 to R10. Higher values will have irreversible side effects on the cells and reduce overall delivery efficiency (Verdurmen and Brock, 2011). Nuclear localization signal (NLS) is a small peptide rich in arginine, lysine or proline commonly found in CPP. NLS can be transported into the cell nucleus through the classical nuclear introduction pathway (Tammam et al., 2017). Due to the limited positive charge and the limited membrane penetration ability of NLS, it is often combined with hydrophobic or amphoteric amino acid sequences to produce effective and versatile amphiphilic vectors including MPG (Lee et al., 2014) and Pep-1 (Yang et al., 2005).
Amphiphilic CPPs contain polar and non-polar amino acid regions, and the non-polar regions are rich in hydrophobic amino acids (for example, alanine, valine, leucine, and isoleucine). Some amphiphilic CPPs are derived entirely from natural proteins such as pVEC, ARF (19–31). ARF (19–31) is from the N-terminal domain of the tumor suppressor gene p14ARF protein (19–31) (Johansson et al., 2008). Chimeric peptides obtained by partially covalently bonding hydrophobic fragments and NLS via amphiphilic CPPs, such as Pep1 (KETWWETWWTEWSQPKKRKV), MPG (GLAFLGFLGAAGSTMGAWSQPKKKRKV) are both based on the SV40 NLS (PKKRKV) (Milletti, 2012).
Cellular Uptake Mechanisms of CPPs
The cellular uptake pathways of CPPs or CPP/cargoes have been generally divided into two types according to whether energy is required or not in the process of internalization (Chugh et al., 2010; Zhu and Jin, 2017): direct translocation and endocytosis.
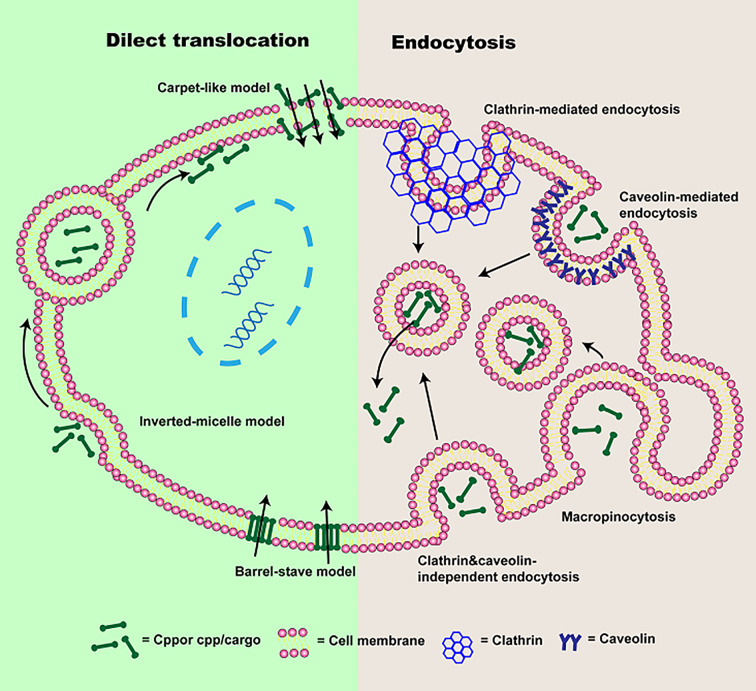
Direct translocation, also known as the non-endocytic uptake pathway, is energy-independent (Reissmann, 2014; Tashima, 2017; Derakhshankhah and Jafari, 2018). It occurs initially through electrostatic interaction or hydrogen bonding between phospholipid bilayer and CPPs or CPP/cargoes (Zhu and Jin, 2017; Pescina et al., 2018). The interaction is followed by CPPs or CPP/cargoes entrance via pore formation or membrane destabilization (Guidotti et al., 2017). According to the different transduced mechanisms, direct translocation is mainly divided into three models: “Barrel-Stave” model (Munjal et al., 2017), “Carpet-like” model (Guidotti et al., 2017) and Inverted-micelle model.
It has been shown that energy-dependence endocytosis is the prevailing cellular uptake mechanism for large molecules weight CPPs or CPP/cargoes (Borrelli et al., 2018; Yang et al., 2019a). So far, four different pathways including macropinocytosis (Wadia et al., 2004), caveolin-mediated endocytosis (Fittipaldi et al., 2003), clathrin-mediated endocytosis (Yang et al., 2016), and clathrin- and caveolin-independent endocytosis (Yang et al., 2019a) have been used to describe endocytosis.
Application of CPPs in the Diagnosis and Treatment of Various Diseases
CPPs application in preclinical studies has obtained great achievements, demonstrated the boundless potential of CPPs-based therapies. It is disappointing that no CPPs-based drugs have been approved by the FDA.
| Compound | CPPs | Cargos | Organization | Therapeutic application | Status | Effect | ClinicalTrials.gov ID | Refs |
|---|---|---|---|---|---|---|---|---|
| AM-111 | TAT | D-JNKI-1 | Auris Medical, Inc. | Acute Inner Ear Hearing Loss | Phase 3 | AM-111 exhibited effective otoprotection in idiopathic sudden sensorineural hearing loss after acute cochlear injury | NCT02561091 Completed 2017 | (Staecker et al., 2019) |
| P28GST | P28 | Glutathione-S-transferase | University Hospital, Lille | Crohn’s disease patients | Phase 2 | P28GST induced slight changes of overall fecal bacterial composition in Crohn’s disease patients | NCT02281916 Completed 2018 | (Capron et al., 2019) |
| P28 | P28 | non-HDM2-mediated Peptide Inhibitor of p53 | Pediatric Brain Tumor Consortium | Central Nervous System Tumors | Phase 1 | Data demonstrated that phase II adult recommended dose of p28 is well-tolerated for children with recurrent CNS malignancies. | NSC745104 Completed 2017 | (Lulla et al., 2016) |
| P28 | P28 | P28 | CDG Therapeutics, Inc. | Solid Tumors That Resist Standard Methods of Treatment | Phase 1 | No Study Results Posted | NCT00914914 Completed 2017 | ClinicalTrials.gov |
| XG-102 | TAT- | dextrogyre peptide | Xigen SA | Postoperative Ocular Inflammation | Phase 3 | Ocular inflammation postoperative effect of XG-102 with a single subconjunctival injection after ocular surgery was lower than dexamethasone eye drops | NCT02508337 Completed 2017 | (Chiquet et al., 2017) |
| DTS-108 | a highly charged oligopeptide of human origin | SN38 | Drais Pharmaceuticals, Inc. | Tumor | Phase 1 | Advanced or metastatic solid tumors patients could receive a DTS-108 dose of 313 mg/m2 every 2 weeks via intravenous. The maximum tolerated dose of DTS-108 was 416 mg/m2. | NA | (Coriat et al., 2016) |
| AVB-620 | ACPPs | Cy5 and Cy7 | Avelas Biosciences, Inc. | Tumor imaging | Phase 1 | AVB-620 improved intraoperative cancer visualization with high safety. NOAEL in rats with single-dose was more than 110-fold of human clinical application dose. | NCT02391194 Completed 2017 |
Gao et al. discovered a novel highly hydrophobic cyclic CPPs (Cyclosporin A, CsA) with electronic neutral, which showed several folds higher penetrating capacity than PFV (PFVYLI) and pentapeptide VPT (VPTLQ) in MCF-7 cells, it was significantly more effective than conventional neutral CPPs. Efficiency and toxicity of cyclosporin A were compared to TAT by delivering a membrane-impenetrable pro-apoptotic peptide (PAD). When CsA conjugated to PAD, the uptake of PDA was improved 2.2- to 4.7-fold in the tumor cell lines tested by CsA, and cellular uptake of CsA-PAD was generally greater than TAT-PAD. Cytotoxicity of CsA-PAD was similar or greater than TAT-PAD in four different tumor cell lines, which depended on the cell type, but it was significantly stronger than PAD.
Recently developed on CPPs application in central nervous system disorders.
| CPPs | Cargos | Delivery platform/stimuli-responsive | Disease | Model | Effect | Ref |
|---|---|---|---|---|---|---|
| CAMP | human metallothionein 1A (hMT1A) | Fusion protein | Parkinson’s disease | A mouse model of PD | CAMP could deliver cargo to mitochondria to alleviate mitochondrial damage | (Kang et al., 2018) |
| TP10 | dopamine | Fusion protein | Parkinson’s disease | Preclinical animal model of PD | TP10-dopamine form accessed to the brain tissue and showed significant anti-parkinsonian activity | (Rusiecka et al., 2019) |
| TAT | ND-13 | Fusion protein | Parkinson’s disease | Mouse model with DJ-1 knockout | TAT modified ND-13 improved the behavioral outcome and dopaminergic system dysfunction | (Finkelstein et al., 2015) |
| MAP | Rasagiline (RAS) | Prodrug | Parkinson’s disease | A human synucleinopathy cell model | RAS-MAP reduced protein alpha-synuclein in cells | (Vale et al., 2020) |
| R9 | amyloid | Fusion protein | Alzheimer’s disease | Cellular level | Total Tau decreased | (Veloria et al., 2017) |
| RVG-9R | BACE1 siRNA | Chitosan-coated solid lipid nanoparticles | Alzheimer’s disease | Cellular level | Prolong residence time in the nasal cavity and improve Nose-to-brain delivery of siRNA | (Rassu et al., 2017) |
| K16ApoE | Curcumin | Target nanoparticles | Alzheimer’s disease | Cellular level and in vivo distribution | Nanoparticles could specifically accumulate in brain vasculature and also detect brain amyloid plaques. | (Ahlschwede et al., 2019a) |
| Penetratin | Ru(II) complex | Ru@Pen@PEG-AuNS | Alzheimer’s disease | Cellular level and in vivo distribution | Ru@Pen@PEG-AuNS could obviously inhibit the formation of Aβ fibrils, BBB permeability was significantly increased | (Yin et al., 2016) |
| R9 | Cy5 | ACPPs, ACPPs dendrimer/MMPs | Stroke | Cellular level and in vivo distribution | ACPPs could response MMP-2/-9 | (Chen et al., 2016b) |
| R9 | NA | Fusion protein | Stroke | Rat stroke model | Poly-arginine exhibited highly neuroprotective | (Meloni et al., 2015) |
Limitations of CPP-Mediated Application
So far, no CPP-conjugated drugs have been approved by FDA and several clinical trials have been terminated. The reasons are as follows: (1) Stability tissues in vivo (Pujals and Giralt, 2008). The rapid clearance from blood is a drawback as a therapeutic payload may be degraded in circulation before reaching the therapeutic site caused by enzymic degradation. (2) Immunogenicity issues. CPPs, due to its polypeptide property, will increase the risk of the undesired immune response in patients, which may not only reduce the drug effect but also cause unwanted immune stress response (Jarver et al., 2010). It can be used in high-dose and frequent intervals with the purpose of increasing exposure to reduce the immunogenicity of therapeutics, but this way results in toxicity. The other way is to inject therapeutics subcutaneously which can reduce the production of anti-drug antibodies caused by the immune system (Jauset and Beaulieu, 2019). (3) Cellular toxicity. Due to off-target cellular absorption of the therapeutics by normal tissues, CPPs can be internalized by almost all types of cells. Most research reported low toxicity of CPPs, however, it should be noticed that everything can become cytotoxic at a high concentration, and many questions need to be answered before being officially applied to the patients. The cytotoxicity of CPPs is of great concern (Saar et al., 2005). For example, the MAP, as a typical CPP, is similar in the structure of the antimicrobial lytic peptides to affect microbial cells by disturbing their plasma membranes (Zorko and Langel, 2005). It has been reported that the MAP showed a fairly high toxic effect on various cell types at concentrations higher than 1 μM through carrying out a variety of cytotoxicity assays (Aguilera et al., 2009). Due to their amphipathic effect in the presence of artificial micelles, MAPs can induce the leakage of protons, proteins, metal ions, etc., which results in cell death due to the damage of the plasma membrane (Moutal et al., 2015). (4) Low specificity. It is well accepted that cationic CPPs can bind to glycosaminoglycans (Walrant et al., 2017), but it is an unknown field whether CPPs can interact with specific membrane receptors. A widespread tissue distribution of CPP-conjugated therapeutics can reduce drug efficiency due to a lower local concentration.
Endosomal Escape Efficiency
The “proton sponge” effect is also used to enhance the endosomal release of CPPs. When the pH of lysosome decreases, the buffering capacity of an agent can capture a large number of protons and cause Cl-influx, which results in lysosome osmotic swelling, and finally lysosome rupture to release the internalized CPP/cargo complexes to the cytoplasm. Another commonly used agent is histidine. The imidazole group of histidine can be protonated to cause lysosome osmotic swelling and rupture of the endosomes (Beloor et al., 2015), which has been extensively used to heighten the gene expression of a TAT/pDNA complex (Lo and Wang, 2008). Another effective way is to use membrane-disruptive peptides to improve the endosomal release of CPPs (Wadia et al., 2004). As we know, viruses can overcome the endosomal trap easily, so using the mechanism of the virus to realize endosomal escape is feasible by conjugating a viral fusion sequence to CPP/cargo complexes (El-Andaloussi et al., 2006). HA2 peptide originated from the hemagglutinin protein of influenza virus is a pH-sensitive fusogenic peptide. The HA2 peptide has an α-helix structure at its N-terminus, which can be inserted into lipids. At the low pH environment inside endosomes, a conformational change exposes the α-helix structure to fuse with the endosomal lipids, resulting in the endosomal release of complexes with proteins and transportan–peptide nucleic acid (PNA) complexes in the cytosol (Trabulo et al., 2010).